|
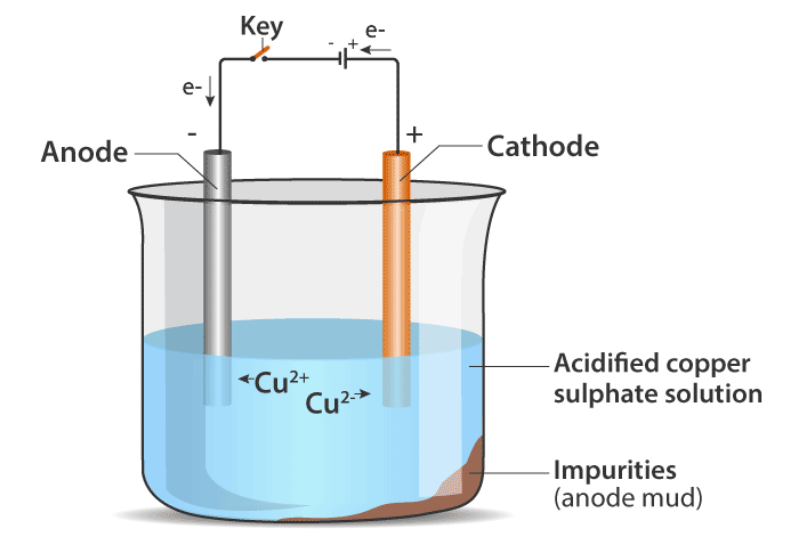
Carbon cannot reduce the oxides of sodium, magnesium, calcium, aluminium, etc., to the respective metals. This is because these metals have more affinity for oxygen than carbon. These metals are obtained by electrolytic reduction.
For example, sodium, magnesium and calcium are obtained by the electrolysis of their molten chlorides. The metals are deposited at the cathode (the negatively charged electrode), whereas, chlorine is liberated at the anode (the positively charged electrode). The reactions are : At cathode (Na⁺) + (e⁻) → Na ; At anode (2Cl⁻) → Cl2 + (2e⁻) Similarly, aluminium is obtained by the electrolytic reduction of aluminium oxide.
0 Comments
I am really happy to share the DAV Question Papers (Bihar Zone) for the Half Yearly Exam Class 10 Session 2023-24 that you have been waiting for. These papers help you do really well in your CBSE exams. So, don't miss out on this opportunity to boost your exam preparation. Head over to the JSUNIL TUTORIAL website now and access these DAV Question /Sample Papers for Half Yearly Exam 2023-24 class 10 session 2023-24. Together, we can achieve excellence in academics. Happy learning and best wishes for your exams! Science Question Paper 2023-24 DAV school, samastipur Download Maths Question Paper 2023-24 DAV school, samastipur Download English Question Paper 2023-24 DAV school, samastipur Download Coming soon ........ Question paper Poddar school samastipur Sample Papers Half Yearly Exam 2023-24 Class 10 Science Question Paper 2023-24 DAV Public School Bina,UP ENG HINDI MATHS S.S.T SCIENCE Sanskrit sample paper class 10 2023-24 class 10 maths standard question paper 2023 pdf sample paper class 10 2024 sample paper class 10 2023 solutions cbse class 10 standard maths question paper 2023 with solutions maths question paper class 10 2023 cbse class 10 question paper 2023 pdf download Let us discuss about a question related to Physics of class 9 Science book chapter gravitation if earth attract moon by gravitational force, then why moon do not move towards the earth? @jsuniltutorial.in  Satellite of mass m1 orbiting Earth at an altitude r. The centripetal force experienced by the satellite is due to the gravitational force acting on it. When an object, like a satellite, is in a circular orbit, gravity is the only force acting on it, which means that the centripetal force must be equal to the gravitational force: FC = FG From this equation we can calculate the velocity that the satellite needs to stay in orbits around the Earth: FC = m v2/r = FG = G mM/r2 → v = sqrt (G M/r) where m is the mass of the satellite and M is the mass of the Earth. In short answer is: Centripetal force of moon and Gravitational pull of earth balance each other . Let us understand in detail : Gravity is a fundamental force that exists between all objects with mass. In the case of the Earth and the Moon, both exert gravitational forces on each other due to their masses.
This gravitational attraction does indeed cause the Moon to move, but it also has a significant sideways motion. When an electric current is passed through concentrated sodium chloride solution, hydrogen gas forms at the negative electrode, chlorine gas forms at the positive electrode, and a solution of sodium hydroxide also forms.
You might have expected sodium metal to be deposited at the negative electrode. However, sodium is too reactive for this to happen so hydrogen is given off instead. During electrolysis: • hydrogen ions H+(aq) (from the water) are discharged at the negative electrode as hydrogen gas, H2(g) • chloride ions Cl–(aq) (from the dissolved sodium chloride) are discharged at the positive electrode as chlorine gas, Cl2(g) • sodium ions Na+(aq) (from the dissolved sodium chloride) and hydroxide ions OH–(aq) (from the water) stay behind - they form sodium hydroxide solution, NaOH(aq) • At the cathode (negative electrode), the H+ cations are reduced when they gain electrons: 2H+(aq) + 2e- → H2(g) • At the anode (positive electrode), the Cl- anions are oxidised when they lose electrons: 2Cl-(aq) → Cl2(g) + 2e- Activity 2.1 NCERT Chapter 2 Metals and Non Metals
Indicator Acid Base Red litmus No Change Blue Blue Litmus Red No change Phenolphthalein Colourless Pink Methyl Orange Pink Yellow whether a substance is acidic or basic by Indictors are substance which change colour in acidic or basic media. Activity 2.2 There are some substances whose odour changes in in acidic or basic media. These are called olfactory indicators. Like onion vanilla, onion and clove. These changes smell in basic solution. Activity 2.3 Take about 5 mL of dilute sulphuric acid in a test tube and add few pieces of zinc granules to it. => You will observe bubbles of hydrogen gas on the surface of zinc granules. Zn + H2SO4 -- >. ZnSO4 + H2 => Pass the Hydrogen gas through the soap solution. Bubbles formed in the soap solution as Hydrogen gas it does not get dissolved in it. Take a burning candle near a gas filled bubble. Candle burn with pop sound. The metal combines with the acid and forms a compound called a salt. Read full post NCERT Activity 2.1 to 2.14 |
Blog SeaRCH Link
All
Join Us For Update |